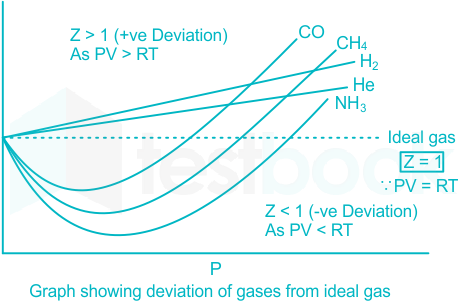
Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
$ 7.00 · 4.6 (590) · In stock

Compressibility factor Z = PV / nRT is plotted against pressure as shown below:What is the correct order for the liquefiability of the gases shown in the above graph? A. CO 2
Compressibility factor Z - PV - nRT is plotted against pressure as shown below-What is the correct order for the liquefiability of the gases shown in the above graph- A- CO 2- CH 4- N 2- H 2B- H 2- CH 4- N 2- CO 2C- CH 4- H 2- N 2- CO 2D- H 2- N 2- CH 4- CO 2

Compressibility factor Z - Gaseous State
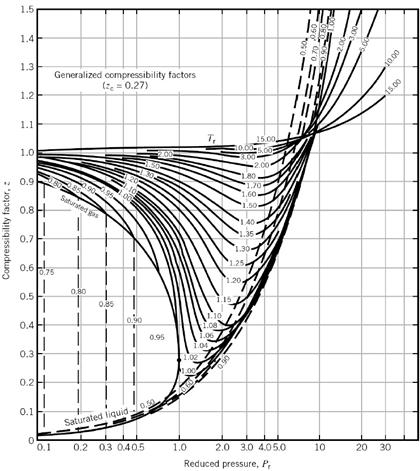
Z= PVm / RT for gases as a function of the reduced

CO2 Z =1 What is the correct increasing order of liquifiability of
Why does gas liquefy at high pressure? Even at high-pressure

PV Compressibility factor Z= nRT is plotted against pressure : N
Solved] The compressibility factor for an ideal gas is

y factor Compressibility factor 2 V is plotted agalnst pressure RT

gaseous state

gaseous state

The given graph represent the variations of Z (compressibility